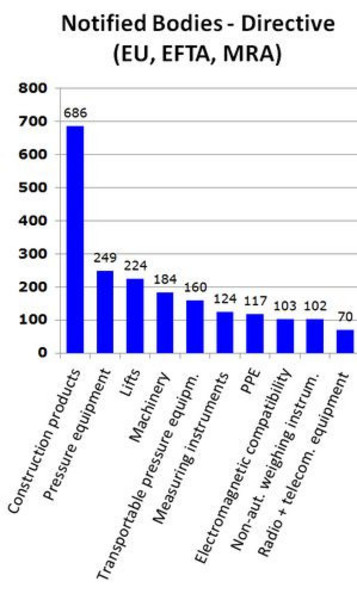
MDR: 26 Notified Bodies on NANDO & Swiss economic operator's requirements updated! · MDlaw – Information platform on European medical device regulations
FFP2 masks and certification: how to carry out a self-check - Mascherine chirurgiche ed FFP2 - iVision Health
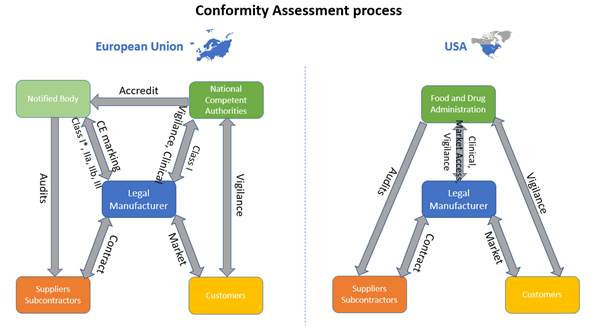
What are the principal differences between the conformity assessment process of a medical device in the USA and in the European Union? - Kvalito

Mario Gabrielli Cossellu on LinkedIn: #notifiedbody #ivdr #nando #mdr #conformityassessment #regulations…

Mario Gabrielli Cossellu on LinkedIn: #notifiedbody #mdr #nando #ivdr #conformityassessment #regulations…

Designation process of MDR/IVDR Notified Bodies - update · MDlaw – Information platform on European medical device regulations